Through the Glass: A Practitioner's Guide to Spectroscopic Windows, Cells & Optics
The glass between your light source and your detector is never neutral. Every optical material has a transmission window, a refractive index, a hardness, and a price — and choosing the wrong one can silently corrupt your data or simply make your instrument impossible to maintain.
When spectroscopists discuss instruments, the conversation almost always centres on detectors, sources, and chemometrics. The optical window — the material that confines, presents, or transmits the sample — is treated as a passive component. It is not. The choice of glass, crystal, or polymer for your cuvette, sample cup, or fibre probe tip determines which wavelengths actually reach your sample, how much signal you lose to reflection and absorption, whether you can clean the surface without scratching it, and whether the material itself contributes spectral features that your calibration must work around.
This guide covers the principal optical materials used in spectroscopic sample containment and windows — their transmission ranges, mechanical and chemical properties, where they genuinely excel, and where they fail. Understanding these materials is not an academic exercise; it is a prerequisite for designing spectroscopic measurements that are physically sound.
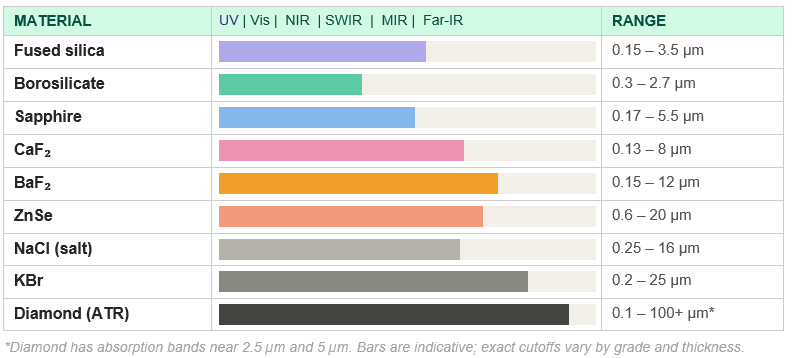
Transmission windows at a glance
Every optical material has a characteristic transmission window — the wavelength range across which it is sufficiently transparent to be useful. The table below maps the approximate useful transmission range for each major spectroscopic optical material, from UV through the far-infrared.
The materials in depth
Understanding each material requires looking beyond its transmission range. Hardness, solubility, refractive index, thermal stability, and cost all determine whether a material is practical — not just optically suitable — for a given application.
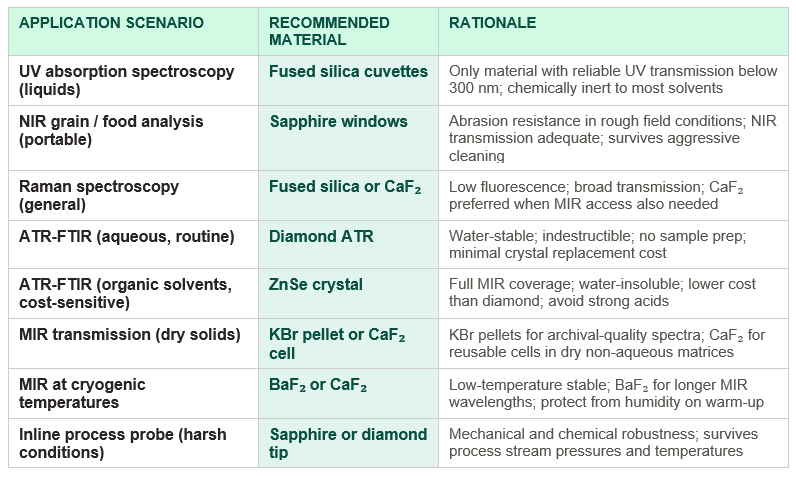
Choosing by application: a decision guide
The refractive index factor
Every optical surface reflects some fraction of light — and that fraction depends on refractive index. High-index materials like ZnSe (~2.4) and germanium (~4.0) lose 15–30% of light per surface through Fresnel reflection. For ATR, this is exploited to generate the evanescent wave. For transmission cells, it represents a signal loss that must be managed with anti-reflection coatings or accounted for in baseline measurements. Low-index materials like CaF₂ (~1.43) and BaF₂ (~1.47) lose only 3–4% per surface — a significant advantage in weak-signal or low-concentration applications.
Practical considerations often overlooked
Transmission range and hardness are the headline specifications, but several second-order factors regularly trip up practitioners making material selections.
Autofluorescence is one. Standard borosilicate glass, and to a lesser extent even fused silica, can fluoresce under laser excitation — a particular problem in Raman spectroscopy where the fluorescence background can swamp the Raman signal. CaF₂ and fused silica of optical grade show very low autofluorescence and are the standard choices for Raman cells.
Birefringence matters in polarised spectroscopy. Materials with cubic crystal symmetry — CaF₂, BaF₂, ZnSe, NaCl — are optically isotropic. Sapphire (hexagonal) and quartz (trigonal) are birefringent: they split polarised light into two components with different refractive indices. For polarimetry and some Raman configurations, this makes sapphire and crystalline quartz unsuitable; fused (amorphous) silica does not share this limitation.
Window thickness is an underappreciated variable. A thicker window absorbs more light within the material’s own absorption bands, compressing the usable spectral range. For materials like BaF₂ and ZnSe operating near their transmission limits, window thickness directly constrains the useful wavelength range — thinner windows push the cutoff further.
Selecting the right glass
No single optical material dominates spectroscopy — each occupies a niche defined by the intersection of its transmission window, mechanical properties, chemical compatibility, and cost. Fused silica owns the UV-to-NIR range for robust, clean measurements. Sapphire extends performance into abrasive and demanding field environments. CaF₂ bridges UV and MIR with low fluorescence. ZnSe handles aqueous ATR affordably. Diamond does everything within its transmission window, indestructibly, at a price. Understanding these trade-offs is not optional for the spectroscopist — it is the difference between a measurement and a mistake dressed up as data.