Into the Dark: Why NIRS Struggles with Absorbent Samples — and How We Fix It
Dark, highly absorbent matrices are among the most persistent challenges in near-infrared spectroscopy. Understanding the physics of why they fail reveals exactly what needs to change — in instrument geometry, sampling strategy, and sample containment.
Near-infrared spectroscopy is a powerful, non-destructive analytical technique — fast, reagent-free, and capable of simultaneously quantifying multiple chemical properties from a single scan. Yet every practitioner encounters the same wall eventually: the sample that simply will not cooperate. Dark samples — heavily pigmented materials, high-carbon matrices, charred residues, dark soils, black plastics, certain food products — absorb so much incident light that the reflectance signal reaching the detector is too weak or too noisy to be analytically useful. This is not a calibration problem. It is a physics problem. And solving it requires addressing the root cause, not papering over it with better chemometrics.
The physics of why dark samples are so difficult
NIRS in diffuse reflectance mode works by directing near-infrared radiation at a sample surface and collecting the scattered light that re-emerges. The detector sees two overlapping signals: specular reflectance (surface glare, chemically uninformative) and diffuse reflectance (light that has penetrated the sample, interacted with chemical bonds, and scattered back out). It is the diffuse component that carries the analytical information.
In dark samples, broadband chromophores — melanins, polyphenolic complexes, carbon black, iron oxides, chlorophyll degradation products — absorb heavily across the NIR range. The light that penetrates even a short distance is largely annihilated before it can scatter back to the detector. The result is very low absolute reflectance values (often below 5%), which compresses the dynamic range available for chemical discrimination. Noise, which is a fixed floor, now represents a much larger fraction of the signal. The signal-to-noise ratio collapses.
Compounding this, dark samples often show non-linear responses to concentration changes. Because so little light returns, small variations in sample packing, surface roughness, or particle size produce reflectance changes that rival or exceed the chemical signal of interest. The analyte and the physical artifact become indistinguishable.
Four failure modes in practice
Addressing the problem: better measurement strategies
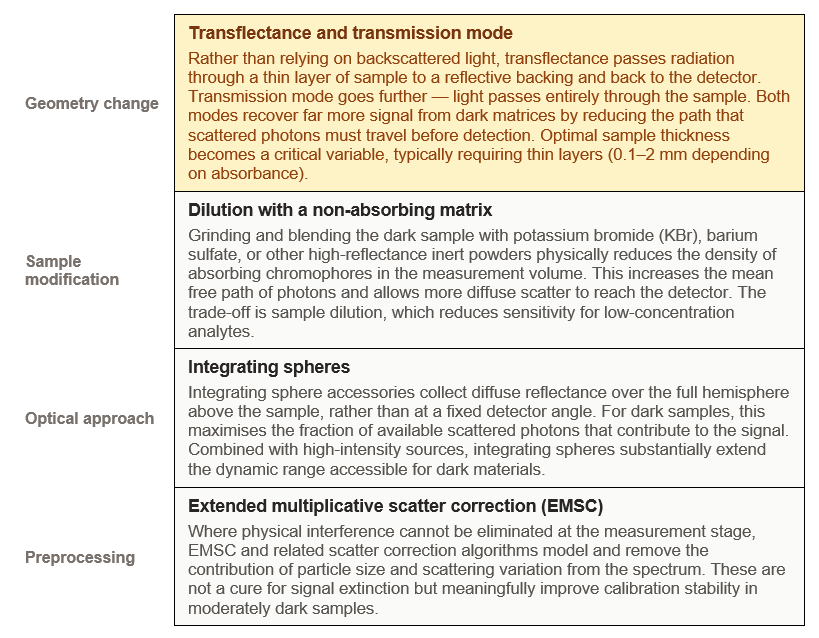
The solutions fall into three broad categories: changing the measurement geometry, modifying the sample itself, or switching to a complementary mode of NIR measurement. In most industrial and laboratory settings, a combination of approaches is necessary.
The wavelength selection angle
Not all NIR wavelengths penetrate dark samples equally. Shorter NIR wavelengths (700–1100 nm) are more susceptible to absorption by dark chromophores than longer wavelengths (1100–2500 nm). Selecting wavelength regions with better penetration depth — or weighting calibration models toward less-absorbed regions — can partially mitigate dark sample losses without changing the sample or geometry.
The underappreciated role of sample cups and optical windows
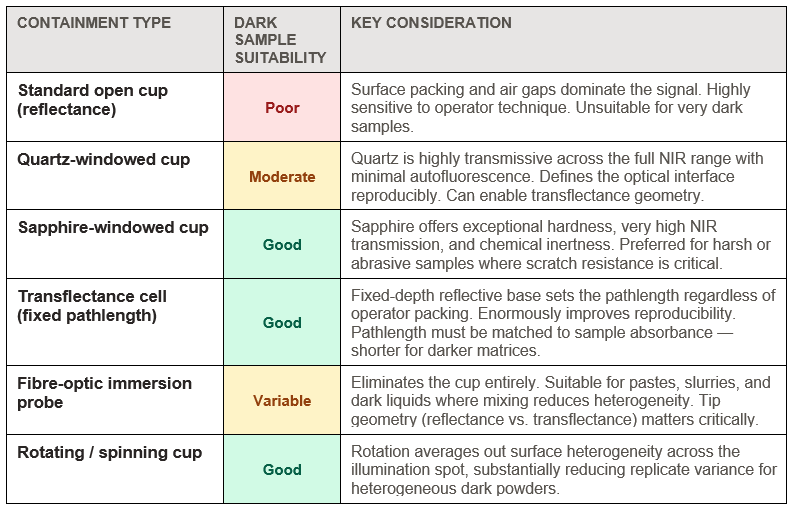
Perhaps the most overlooked variable in dark sample NIRS is the sample containment itself. The choice of cup material, window geometry, and window optical properties directly determines how much light reaches the sample, how the measurement geometry is defined, and whether surface effects dominate the measurement.
The window material is not a passive optical element — it defines the first interface that light encounters. A scratched or contaminated window introduces stray scatter that is indistinguishable from sample signal. For dark samples where the sample signal is already low, window contributions become proportionally significant. Routine inspection and replacement of optical windows is a basic but frequently neglected quality control step.
Cup geometry also matters for another reason: it determines how reproducibly the sample is presented to the instrument. For dark, heterogeneous powders (dark soils, biochar, dark feed ingredients), an open cup allows the analyst's tamping pressure to vary the packing density between measurements — and packing density directly affects the depth of light penetration and therefore the effective sample volume interrogated. Fixed-depth cells with a defined reflective base remove this variable entirely.
Complementary techniques when NIRS reaches its limit
Sometimes the honest answer is that diffuse reflectance NIR is not the right tool for a given dark sample. Raman spectroscopy, which detects inelastic scattering rather than reflectance, is often less sensitive to dark sample absorbance — though fluorescence interference becomes the competing problem. Mid-infrared spectroscopy in attenuated total reflectance (ATR-MIR) interrogates only the surface layer of a sample, making penetration depth irrelevant, and can be highly effective for dark pastes and films. X-ray fluorescence addresses elemental composition in cases where NIR is chasing the wrong signal entirely. Recognising when to reach for a complementary technique is part of sound spectroscopic practice. NIR's speed and non-destructiveness are genuine advantages — but they do not override physics.
Practical guidance
Dark samples fail in NIRS because signal extinction outpaces detector sensitivity, and physical heterogeneity swamps chemical information. The path forward is systematic: switch to transflectance or transmission geometry where possible; use sapphire or quartz-windowed fixed-depth cells to define the optical interface and remove packing variability; consider dilution with an inert matrix for extreme cases; and apply scatter correction in preprocessing. When the sample is so dark that no NIR geometry yields an adequate SNR, that is not a calibration failure — it is a signal to reach for a different technique.